|
Bip, a member of the heat shock protein 70 family ( HSP70), promotes proper protein folding. Proteins synthesized in the rough endoplasmic reticulum fold correctly with the help of endoplasmic reticulum lumen chaperone BiP and are packaged into transport vesicles for transport to the Golgi apparatus. After the signal peptide enters the endoplasmic reticulum, it is cut off from the peptide chain by signal peptidase and quickly degraded by other proteases. The transposon is a channel containing several Sec proteins ( Sec61, Sec62/ Sec63) across the lipid double membrane. The signal peptide guides the new peptide to enter the rough endoplasmic reticulum lumen through transposon co-translation. At the same time, SRP is released into the cytoplasmic matrix and rebinds to other signal peptides. After that, the SRP-ribosome complex moves to the endoplasmic reticulum and binds to the SRP receptor on the endoplasmic reticulum, restarting the extension of the peptide chain. Once SRP binds to ribosomes with new peptides, the extension of the peptide chain is terminated. The translation of secretory or integrin proteins initiates in the cytoplasm, and the ribosomes containing these mRNAs are identified by signal recognition particles (SRPs) through a signal sequence called signal peptides within the amino terminus of the newly synthesized peptides. Nascent peptides pass through the endoplasmic reticulum and enter the endoplasmic reticulum lumen in the rough endoplasmic reticulum. Other proteins, such as secretory proteins, integral membrane proteins, and soluble resident proteins in organelles, are transferred to the endoplasmic reticulum along with ribosomes shortly after the initial synthesis. Some proteins are produced on free ribosomes in the cytoplasm, which remain in the cytoplasmic matrix or are transported to the nucleus, mitochondria, and peroxidases. Protein Processing in the Endoplasmic Reticulum When proteins are improperly processed or modified, the endoplasmic reticulum acts as a quality control agent, trapping the non-folded or misfolded proteins in the endoplasmic reticulum and clearing them through the ubiquitin-proteosome pathway (UPP). The newly synthesized peptides are transported to the endoplasmic reticulum and folded into the correct three-dimensional structure before being transported to their destination. Why Do Proteins Need to Be Processed?įor specific secreted proteins, polypeptide chains need to be modified in different ways post-translation to obtain physiological functions. Cells do not contain pure rough or smooth endoplasmic reticulum they are part of a continuous structure of the endoplasmic reticulum, respectively. Cells that specialize in producing proteins have more rough endoplasmic reticulum, while cells that produce lipids (fats) and steroid hormones have more smooth endoplasmic reticulum. Smooth endoplasmic reticulum also has the function of detoxification and glycogen metabolism. The smooth endoplasmic reticulum synthesizes almost all lipids, including phospholipids and cholesterol. The smooth endoplasmic reticulum is branched tubular or vesicular without ribosome attachment. 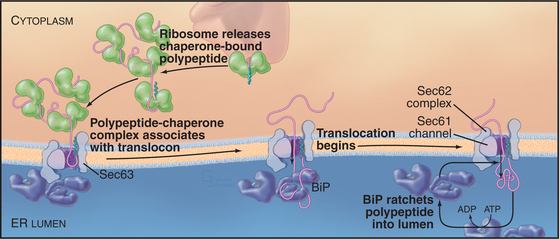
The rough endoplasmic reticulum is a flattened sac, neatly arranged with ribosomes embedded on its surface, which is the site of protein synthesis. There are two types of endoplasmic reticulum: rough endoplasmic reticulum (RER) and smooth endoplasmic reticulum (SER). The endoplasmic reticulum exists in most eukaryotic cells except for mature red blood cells in mammals. The endoplasmic reticulum is a continuous membranous system that forms a network of interconnected vesicles surrounded by flat membranous sacs or tubular structures. observed cultured mouse fibroblasts, they found a network structure in the cytoplasm, which was later named endoplasmic reticulum (ER).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
